Making a Density Tower
Density is a foundational concept in science, which explains why boats float, rocks sink and balloons fly away when you let go of them. With just a few materials from home the boys conducted their own colourful density experiment and saw how this principal works first hand.
This experiment involved forming a density tower using a combination of different liquids that formed layers on top of each other due to the differences in their density.
Key Words
Sink: To go below the surface of the water.
Float: To stay on or above the surface of the water.
Observing: Using our senses to gather information.
Density: How compact the particles of an object are.
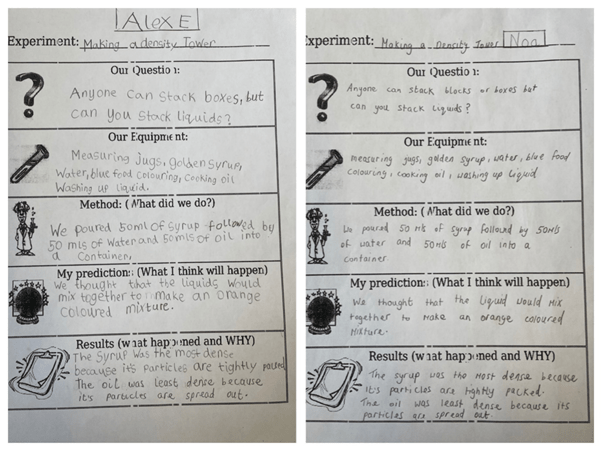
Our Question: Anyone can stack blocks, boxes or books but can you stack liquids?
Materials Used:
Cooking oil
Blue food colouring
Water
Golden Syrup
Measuring jugs.


Method:
We poured 50mls of the golden syrup into a container. Next, we poured 50mls of water coloured with blue food colouring into the container. Lastly, we poured 50mls of cooking oil into the container.








Our Predictions:
We thought that the liquids would mix together to make an orange coloured mixture.
Our Results:

Each of the liquids had a different mass of particles which is the same as having a different amount of tiny parts squashed into them. This makes them have different densities and therefore one can sit on top of the other. The more dense a liquid is, the heavier it is. The golden syrup was the most dense because it’s particles are tightly packed together. It sank to the bottom of the tower. The water floated on top of the syrup. The cooking oil was the least dense because its’ particles are more spread out. It was the lightest liquid so it floated to the top.



